Free Access
| Issue |
Environ. Biosafety Res.
Volume 6, Number 1-2, January-June 2007
Thematic Issue on Horizontal Gene Transfer
|
|
|---|---|---|
| Page(s) | 3 - 13 | |
| DOI | https://doi.org/10.1051/ebr:2007034 | |
| Published online | 26 October 2007 | |
- Adamczyk M, Jagura-Burdzy G (2003) Spread and survival of promiscuous IncP-1 plasmids. Acta Biochim. Pol. 50: 425–453 [PubMed] [Google Scholar]
- Arnold DL, Pitman A, Jackson RW (2003) Pathogenicity and other genomic islands in plant pathogenic bacteria. Mol. Plant Pathol. 4: 407–420 [CrossRef] [PubMed] [Google Scholar]
- Ashelford KE, Day MJ, Fry JC (2003) Elevated abundance of bacteriophage infecting bacteria in soil. Appl. Environ. Microbiol. 69: 285–289 [CrossRef] [PubMed] [Google Scholar]
- Bale MJ, Fry JC, Day MJ (1988) Novel method for studying plasmid transfer in undisturbed river epilithon. Appl. Environ. Microbiol. 54: 2756–2758 [PubMed] [Google Scholar]
- Berg OG, Kurland CG (2002) Evolution of microbial genomes: Sequence acquisition and loss. Mol. Biol. Evol. 19: 2265–2276 [PubMed] [Google Scholar]
- Berka RM, Hahn J, Albano M, Draskovic I, Persuh M, Cui X, Sloma A, Widner W, Dubnau D (2002) Microarray analysis of the Bacillus subtilis K-state: genome-wide expression changes dependent on ComK. Mol. Microbiol. 43: 1331–1345 [CrossRef] [PubMed] [Google Scholar]
- Blum SEA, Lorenz MG, Wackernagel W (1997) Mechanisms of retarded DNA degradation and prokaryotic origin of DNase in nonsterile soils. Syst. Appl. Microbiol. 20: 513–521 [Google Scholar]
- Boon N, Goris J, De Vos P, Verstraete W, Top EM (2001) Genetic diversity among 3-chloroaniline- and aniline-degrading strains of the Comamonadaceae. Appl. Environ. Microbiol. 67: 1107–1115 [CrossRef] [PubMed] [Google Scholar]
- Brüssow H, Canchaya C, Hardt W (2004) Phages and the evolution of bacterial pathogens: from genomic rearrangements to lysogenic conversion. Microbiol. Mol. Biol. Rev. 68: 560–602 [CrossRef] [PubMed] [Google Scholar]
- Brzuszkiewicz E, Brüggemann H, Liesegang H, Emmerth M, Ölschläger T, Nagy G, Albermann K, Wagner C, Buchrieser C, Emody L, Gottschalk G, Hacker J, Dobrindt U (2006) How to become a uropathogen: comparative genomic analysis of extraintestinal pathogenic Escherichia coli strains. PNAS 103: 12879–12884 [CrossRef] [Google Scholar]
- Burrus V, Waldor MK (2004) Shaping bacterial genomes with integrative and conjugative elements. Res. Microbiol. 155: 376–386 [CrossRef] [PubMed] [Google Scholar]
- Canchaya C, Proux C, Fournous G, Bruttin A, Brüssow H (2003) Prophage genomics. Microbiol. Mol. Biol. Rev. 67: 238–276 [CrossRef] [PubMed] [Google Scholar]
- Cérémonie H, Buret F, Simonet P, Vogel TM (2004) Isolation of lightning-competent soil bacteria. Appl. Environ. Microbiol. 70: 6342–6346 [CrossRef] [PubMed] [Google Scholar]
- Cérémonie H, Buret F, Simonet P, Vogel TM (2006) Natural electrotransformation of lightning-competent Pseudomonas sp. strain N3 in artificial soil microcosms. Appl. Environ. Microbiol. 72: 2385–2389 [CrossRef] [PubMed] [Google Scholar]
- Chen I, Dubnau D (2004) DNA uptake during bacterial transformation. Nature Rev. Microbiol. 2: 241–249 [Google Scholar]
- Costa R, Götz M, Mrotzek N, Lottmann J, Berg G, Smalla K (2006a) Effect of site and plant species on rhizosphere community structure as revealed by molecular analysis of different microbial guilds. FEMS Microbiol. Ecol. 56: 236–249 [CrossRef] [PubMed] [Google Scholar]
- Costa R, Salles JF, Berg G, Smalla K (2006b) Cultivation-independent analysis of Pseudomonas species in soil and in the rhizosphere of field-grown Verticillium dahliae host plants. Environ. Microbiol. 8: 2136–2149 [CrossRef] [PubMed] [Google Scholar]
- Dahlberg C, Chao L (2003) Amelioration of the cost of conjugative plasmid carriage in Escherichia coli K12. Genetics 165: 1641–1649 [PubMed] [Google Scholar]
- Dejonghe W, Goris J, Dierickx A, De Dobbeleer V, Crul K, De Vos P, Verstraete W, Top EM (2002) Diversity of 3-chloroaniline and 3,4-dichloroaniline degrading bacteria isolated from three different soils and involvement of their plasmids in chloroaniline degradation. FEMS Microbiol. Ecol. 42: 315–325 [CrossRef] [PubMed] [Google Scholar]
- Demanèche S, Jocteur-Monrozier L, Quiquampoix H, Simonet P (2001a) Evaluation of biological and physical protection against nuclease degradation of clay-bound plasmid DNA. Appl. Environ. Microbiol. 67: 293–299 [CrossRef] [PubMed] [Google Scholar]
- Demanèche S, Kay E, Gourbière F, Simonet P (2001b) Natural transformation of Pseudomonas fluorescens and Agrobacterium tumefaciens in soil. Appl. Environ. Microbiol. 67: 2617–2621 [CrossRef] [PubMed] [Google Scholar]
- de Vries J, Wackernagel W (1998) Detection of nptII (kanamycin resistance) genes in genomes of transgenic plants by marker-rescue transformation. Mol. Gen. Genet. 257: 606–613 [CrossRef] [PubMed] [Google Scholar]
- de Vries J, Wackernagel W (2002) Integration of foreign DNA during natural transformation of Acinetobacter sp. by homology-facilitated illegitimate recombination. Proc. Natl. Acad. Sci. USA 99: 2094–2099 [CrossRef] [Google Scholar]
- de Vries J, Wackernagel W (2004) Microbial horizontal gene transfer and the DNA release from transgenic crop plants. Plant Soil 266: 91–104 [CrossRef] [Google Scholar]
- de Vries J, Heine M, Harms K, Wackernagel W (2003) Spread of recombinant DNA by roots and pollen of transgenic potato plants, identified by highly specific biomonitoring using natural transformation of an Acinetobacter sp. Appl. Environ. Microbiol. 69: 4455–4462 [CrossRef] [PubMed] [Google Scholar]
- Dobrindt U, Hochhut B, Hentschel U, Hacker J (2004) Genomic islands in pathogenic and environmental microorganisms. Nature Rev. Microbiol. 2: 414–424 [CrossRef] [Google Scholar]
- Doolittle WF, Boucher Y, Nesbo LC, Douady CJ, Andersson JO, Roger AJ (2003) How big is the iceberg of which organellar genes in nuclear genomes are but the tip? Phil. Trans. R. Soc. Lond. B 358: 39–58 [CrossRef] [Google Scholar]
- Drønen AK, Torsvik V, Goksøyr J, Top EM (1998) Effect of mercury addition on plasmid incidence and gene mobilising capacity in bulk soil. FEMS Microbiol. Ecol. 27: 381-394 [Google Scholar]
- Dubnau D (1999) DNA uptake in bacteria. Annu. Rev. Microbiol. 53: 217–244 [CrossRef] [PubMed] [Google Scholar]
- Frey J, Bagdasarian M (1989) The molecular biology of IncQ plasmids. In Thomas CM, ed, Promiscuous plasmids of gram negative bacteria, Academic Press, Orlando, USA, pp 79–94 [Google Scholar]
- Gebhard F, Smalla K (1998) Transformation of Acinetobacter sp. strain BD 413 by transgenic sugar beet DNA. Appl. Environ. Microbiol. 64: 1550–1554 [PubMed] [Google Scholar]
- Gebhard F, Smalla K (1999) Monitoring field releases of genetically modified sugar beets for persistence of transgenic plant DNA and horizontal gene transfer. FEMS Microbiol. Ecol. 28: 261–272 [CrossRef] [Google Scholar]
- Ghigo JM (2001) Natural conjugative plasmids induce bacterial biofilm development. Nature 412: 442–445 [CrossRef] [PubMed] [Google Scholar]
- Gillings MR, Holley MP, Stokes HW, Holmes AJ (2005) Integrons in Xanthomonas: A source of species genome diversity. PNAS 102: 4419–1124 [CrossRef] [Google Scholar]
- Götz A, Smalla K (1997) Manure enhances plasmid mobilization and survival of Pseudomonas putida introduced into field soil. Appl. Environ. Microbiol. 63: 1980–1986 [PubMed] [Google Scholar]
- Grozdanov L, Raasch C, Schulze J, Sonnenborn U, Gottschalk G, Hacker J, Dobrindt U (2004) Analysis of the genome structure of the nonpathogenic probiotic Escherichia coli strain Nissle 1917. J. Bacteriol. 186: 5432–5441 [Google Scholar]
- Hacker J, Carniel E (2001) Ecological fitness, genomic islands and bacterial pathogenicity. EMBO reports 2: 376–381 [PubMed] [Google Scholar]
- Hacker J, Kaper JB (2000) Pathogenicity islands and the evolution of microbes. Annu. Rev. Microbiol. 54: 641–679 [CrossRef] [PubMed] [Google Scholar]
- Hacker J, Hentschel U, Dobrindt U (2003) Prokaryotic chromosomes and disease. Science 301: 790–793 [CrossRef] [PubMed] [Google Scholar]
- Hall RM, Collis CM (1995) Mobile gene cassettes and integrons: capture and spread of genes by site-specific recombination. Mol. Microbiol. 15: 593–600 [Google Scholar]
- Hendrickx L, Hausner M, Wuertz S (2003) Natural genetic transformation in monoculture Acinetobacter sp. strain BD413 biofilms. Appl. Environ. Microbiol. 69: 1721–1727 [CrossRef] [PubMed] [Google Scholar]
- Heuer H, Smalla K (2007) Manure and sulfadiazine synergistically increased bacterial antibiotic resistance in soil at least over two months. Environ. Microbiol. 9: 657–666 [CrossRef] [PubMed] [Google Scholar]
- Heuer H, Krögerrecklenfort E, Egan S, van Overbeek L, Guillaume G, Nikolakopoulou TL, Wellington EMH, van Elsas JD, Collard JM, Karagouni AD, Smalla K (2002) Gentamicin resistance genes in environmental bacteria: prevalence and transfer. FEMS Microbiol. Ecol. 42: 289–302 [CrossRef] [PubMed] [Google Scholar]
-
Heuer H, Szczepanowski R, Schneiker S, Pühler A, Top EM, Schlüter A (2004) The complete sequences of plasmids pB2 and pB3 provide evidence for a recent ancestor of the IncP-1
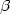 group without any accessory genes. Microbiology 150: 3591–3599
[CrossRef]
[PubMed]
[Google Scholar]
group without any accessory genes. Microbiology 150: 3591–3599
[CrossRef]
[PubMed]
[Google Scholar]
- Heuer H, Fox RE, Top EM (2007) Frequent conjugative transfer accelerates adaptation of a broad-host-range plasmid to an unfavourable Pseudomonas putida host. FEMS Microbiol. Ecol. 59: 738–748 [CrossRef] [PubMed] [Google Scholar]
- Jain R, Rivera MC, Lake JA (1999) Horizontal gene transfer among genomes: The complexity hypothesis. Proc. Natl. Acad. Sci. USA 96: 3801–3806 [CrossRef] [Google Scholar]
- Jain R, Rivera MC, Moore J, Lake JA (2003) Horizontal gene transfer accelerates genome innovation and evolution. Mol. Biol. Evol. 20: 1598–1602 [CrossRef] [PubMed] [Google Scholar]
- Johnsen AR, Kroer N (2006) Effects of stress and other environmental factors on horizontal plasmid transfer assessed by direct quantification of discrete transfer events. FEMS Microbiol. Ecol., DOI:10.1111/j.1574-6941.2006.00230.x [Google Scholar]
- Kobayashi N, Bailey MJ (1994) Plasmids isolated from the sugar beet phyllosphere show little or no homology to molecular probes currently available for plasmid typing. Microbiology 140: 289–296 [CrossRef] [PubMed] [Google Scholar]
- Koonin EV, Makarova KS, Aravind L (2001) Horizontal gene transfer in prokaryotes: quantification and classification. Ann. Rev. Microbiol. 55: 709–742 [Google Scholar]
- Lan L, Reeves PR (2000) Intraspecies variation in bacterial genomes: the need for a species genome concept. Trends Microbiol. 8: 396–401 [CrossRef] [PubMed] [Google Scholar]
- Larraín-Linton J, De la Iglesia R, Melo F, González B (2006) Molecular and population analyses of a recombination event in the catabolic plasmid pJP4. J. Bacteriol. 188: 6793–6801 [CrossRef] [PubMed] [Google Scholar]
- Li YH, Lau PC, Lee JH, Ellen RP, Cvitkovitch DG (2001) Natural genetic transformation of Streptococcus mutans growing in biofilms. J. Bacteriol. 183: 897–908 [CrossRef] [PubMed] [Google Scholar]
- Licht TR, Struve C, Christensen BB, Poulsen RL, Molin S, Krogfelt KA (2003) Evidence of increased spread and establishment of plasmid RP4 in the intestine under sub-inhibitory tetracycline concentrations. FEMS Microbiol. Ecol. 44: 217–223 [CrossRef] [PubMed] [Google Scholar]
- Lilley AK, Bailey MJ (1997) The acquisition of indigenous plasmids by a genetically marked pseudomonad population colonizing the sugar beet phytosphere is related to local environmental conditions. Appl. Environ. Microbiol. 63: 1577–1583 [PubMed] [Google Scholar]
- Lilley AK, Fry JD, Day MJ, Bailey MJ (1994) In situ transfer of an exogenously isolated plasmid between indigenous donor and recipient Pseudomonas spp. in sugar beet rhizosphere. Microbiology 140: 27–33 [CrossRef] [Google Scholar]
- Lilley AK, Bailey MJ, Day MJ, Fry JC (1996) Diversity of mercury resistance plasmids obtained by exogenous isolation from the bacteria of sugar beet in three successive years. FEMS Microbiol. Ecol. 20: 211–227 [CrossRef] [Google Scholar]
- Lorenz MG, Wackernagel W (1994) Bacterial gene transfer by natural genetic transformation in the environment. Microbiol. Rev. 58: 563–602 [PubMed] [Google Scholar]
- Lucchini S, Rowley G, Goldberg MD, Hurd D, Harrison M, Hinton JCD (2006) H-NS mediates the silencing of laterally acquired genes in bacteria. PLoS Pathog. 2: e81, DOI: 10.1371/journal.ppat.0020081 [Google Scholar]
- Maamar H, Dubnau D (2005) Bistability in the Bacillus subtilis K-state (competence) system requires a positive feedback loop. Mol. Microbiol. 56: 615–624 [CrossRef] [PubMed] [Google Scholar]
- Mahillon J, Chandler M (1998) Insertion sequences. Microbiol. Mol. Biol. Rev. 62: 725–774 [Google Scholar]
- Mazel D, Davies J (1999) Antibiotic resistance in microbes. Cell. Mol. Life Sci. 56: 742–754 [CrossRef] [PubMed] [Google Scholar]
- McManus PS, Stockwell VO, Sundin GW, Jones AL (2002) Antibiotic use in plant agriculture. Ann. Rev. Phytopathol. 40: 443 [CrossRef] [Google Scholar]
- Merlin C, Springael D, Toussaint A (1999) Tn4371: A modular structure encoding a phage-like integrase, a Pseudomonas-like catabolic pathway, and RP4-Ti-like transfer functions. Plasmid 41: 40–54 [CrossRef] [PubMed] [Google Scholar]
- Mølbak L, Licht TR, Kvist T, Kroer N, Andersen SR (2003) Plasmid transfer from Pseudomonas putida to the indigenous bacteria on alfalfa sprouts: characterization, direct quantification, and in situ location of transconjugant cells. Appl. Environ. Microbiol. 69: 5536–5542 [CrossRef] [PubMed] [Google Scholar]
- Mølbak L, Molin S, Kroer N (2007) Root growth and exudate production define the frequency of horizontal plasmid transfer in the Rhizosphere. FEMS Microbiol. Ecol. 59: 167–176 [CrossRef] [PubMed] [Google Scholar]
- Nielsen KM, van Weerelt DM, Berg TN, Bones AM, Hagler AN, van Elsas JD (1997) Natural transformation and availability of transforming chromosomal DNA to Acinetobacter calcoaceticus in soil microcosms. Appl. Environ. Microbiol. 63: 1945–1952 [PubMed] [Google Scholar]
- Nielsen KM, Smalla K, van Elsas JD (2000) Natural transformation of Acinetobacter sp. strain BD413 with cell lysates of Acinetobacter sp., Pseudomonas fluorescens, and Burkholderia cepacia in soil microcosms. Appl. Environ. Microbiol. 66: 206–212 [Google Scholar]
- Normander B, Christensen BB, Molin S, Kroer N (1998) Effect of bacterial distribution and activity on conjugal gene transfer on the phylloplane of the bush bean (Phaseolus vulgaris). Appl. Environ. Microbiol. 64: 1902–1909 [PubMed] [Google Scholar]
- Ochman H, Lawrence JG, Groisman EA (2000) Lateral gene transfer and the nature of bacterial innovation. Nature 405: 299–304 [CrossRef] [PubMed] [Google Scholar]
- Paget E, Simonet P (1994) On the track of natural transformation in soil. FEMS Microbiol. Ecol. 15: 109–118 [CrossRef] [Google Scholar]
- Paget E, Simonet P (1997) Development of engineered genomic DNA to monitor the natural transformation of Pseudomonas stutzeri in soil-like microcosms. Can. J. Microbiol. 43: 78–84 [Google Scholar]
- Powell BJ, Purdy KJ, Thompson IP, Bailey MJ (1993) Demonstration of tra+ plasmid activity in bacteria indigenous to the phyllosphere of sugar beet; gene transfer to a recombinant pseudomonad. FEMS Microbiol. Ecol. 12: 195–206 [Google Scholar]
- Pukall R, Tschäpe H, Smalla K (1996) Monitoring the spread of broad host and narrow host range plasmids in soil microcosms. FEMS Microbiol. Ecol. 20: 53–66 [CrossRef] [Google Scholar]
- Reisner A, Höller BM, Molin S, Zechner EL (2006) Synergistic effects in mixed Escherichia coli biofilms: Conjugative plasmid transfer drives biofilm expansion. J. Bacteriol. 188: 3582–3588 [CrossRef] [PubMed] [Google Scholar]
-
Schlüter A, Heuer H, Szczepanowski R, Forney LJ, Thomas CM, Pühler A, Top EM (2003) The 64,508 bp IncP-1
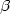 antibiotic multiresistance plasmid pB10 isolated from a wastewater treatment plant provides evidence for recombination between members of different branches of the IncP-1
antibiotic multiresistance plasmid pB10 isolated from a wastewater treatment plant provides evidence for recombination between members of different branches of the IncP-1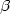 group. Microbiology 149: 3139–3153
[CrossRef]
[PubMed]
[Google Scholar]
group. Microbiology 149: 3139–3153
[CrossRef]
[PubMed]
[Google Scholar]
- Schneiker S, Keller M, Dröge M, Lanka E, Pühler A, Selbitschka W (2001) The genetic organization and evolution of the broad host range mercury resistance plasmid pSB102 isolated from a microbial population residing in the rhizosphere of alfalfa. Nucl. Acids Res. 29: 5169–5181 [CrossRef] [Google Scholar]
- Sengeløv G, Kristensen KJ, Sørensen AH, Kroer N, Sørensen SJ (2001) Effect of genomic location on horizontal transfer of a recombinant gene cassette between Pseudomonas strains in the rhizosphere and spermosphere of barley seedlings. Curr. Microbiol. 42: 160–167 [CrossRef] [PubMed] [Google Scholar]
- Sikorski J, Teschner N, Wackernagel W (2002) Highly different levels of natural transformation are associated with genomic subgroups within a local population of P. stutzeri from soil. Appl. Environ. Microbiol. 68: 865–873 [CrossRef] [PubMed] [Google Scholar]
- Smalla K, Heuer H (2006) How to assess the abundance and diversity of mobile genetic elements in soil bacterial communities? In Nannipieri P, Smalla K, eds, Nucleic acids in soil, Springer Berlin Heidelberg New York, pp 313–330 [Google Scholar]
- Smalla K, Sobecky PA (2002) The prevalence and diversity of mobile genetic elements in bacterial communities of different environmental habitats: insights gained from different methodological approaches. FEMS Microbiol. Ecol. 42: 165–175 [CrossRef] [PubMed] [Google Scholar]
- Smalla K, Heuer H, Götz A, Niemeyer D, Krögerrecklenfort E, Tietze E (2000) Exogenous isolation of antibiotic resistance plasmids from piggery manure slurries reveals a high prevalence and diversity of IncQ-like plasmids. Appl. Environ. Microbiol. 66: 4854-4862 [CrossRef] [PubMed] [Google Scholar]
- Smalla K, Wieland G, Buchner A, Zock A, Parzy J, Kaiser S, Roskot N, Heuer H, Berg G (2001) Bulk and rhizosphere soil bacterial communities studied by denaturing gradient gel electrophoresis: plant-dependent enrichment and seasonal shifts revealed. Appl. Environ. Microbiol. 67: 4742–4750 [CrossRef] [PubMed] [Google Scholar]
-
Smalla K, Haines AS, Jones K, Krögerrecklenfort E, Heuer H, Schloter M, Thomas CM (2006) Increased abundance of IncP-1
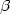 plasmids and mercury resistance genes in mercury polluted river sediments - first discovery of IncP-1
plasmids and mercury resistance genes in mercury polluted river sediments - first discovery of IncP-1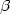 plasmids with a complex mer transposon as sole accessory element. Appl. Environ. Microbiol. 72: 7253–7259
[CrossRef]
[PubMed]
[Google Scholar]
plasmids with a complex mer transposon as sole accessory element. Appl. Environ. Microbiol. 72: 7253–7259
[CrossRef]
[PubMed]
[Google Scholar]
- Smets BF, Barkay T (2005) Horizontal gene transfer: Perspectives at a crossroads of scientific disciplines. Nature Rev. Microbiol. 3: 675–678 [CrossRef] [Google Scholar]
- Smit E, Wolters A, van Elsas JD (1998) Self-transmissible mercury resistance plasmids with gene-mobilizing capacity in soil bacterial populations: influence of wheat roots and mercury addition. Appl. Environ. Microbiol. 64: 1210–1219 [PubMed] [Google Scholar]
- Sørensen SJ, Bailey M, Hansen LH, Kroer N, Wuertz S (2005) Studying plasmid horizontal transfer in situ: a critical review. Nature Rev. Microbiol. 3: 700–710 [CrossRef] [Google Scholar]
- Springael D, Ryngaert A, Merlin C, Toussaint A, Mergeay M (2001) Occurrence of Tn4371 related mobile elements and sequences in PCB degrading bacteria. Appl. Environ. Microbiol. 67: 42–50 [CrossRef] [PubMed] [Google Scholar]
- Tauch A, Schneiker S, Selbitschka W, Pühler A, van Overbeek L, Smalla K, Thomas CM, Bailey MJ, Forney LJ, Weightman A, Ceglowski P, Pembroke A, Tietze E, Schröder G, Lanka E, van Elsas JD (2002) The complete nucleotide sequence and environmental distribution of the cryptic, conjugative, BHR plasmid pIPO2 isolated from bacteria of the wheat rhizosphere. Microbiology 148: 637–653 [Google Scholar]
- Thomas CM (2000) Paradigms of plasmid organization. Mol. Microbiol. 37: 485–491 [CrossRef] [PubMed] [Google Scholar]
- Timms-Wilson TM, van Overbeek LS, Bailey MJ, Trevors JT, van Elsas JD (2001) Quantification of gene transfer in soil and the rhizosphere. In Hurst CJ, Crawford RL, Knudsen GR, McInerney MJ, Stetzenbach LD, eds, Manual of environmental microbiology, ASM Press, Washington, USA, pp 648–659 [Google Scholar]
- Toleman MA, Bennett PM, Walsh TR (2006) Common regions e.g. orf513 and antibiotic resistance: IS91-like elements evolving complex class 1 integrons. J. Antimirob. Chemother. 58: 1–6 [CrossRef] [Google Scholar]
- Top EM, Springael D (2003) The role of mobile genetic elements in bacterial adaptation to xenobiotic organic compounds. Curr. Opin. Biotechnol. 14: 262–269 [CrossRef] [PubMed] [Google Scholar]
- Top EM, Holben WE, Forney LJ (1995) Characterization of diverse 2,4-dichlorophenoxyacetic acid-degradative plasmids isolated from soil by complementation. Appl. Environ. Microbiol. 61: 1691–1698 [PubMed] [Google Scholar]
- Top EM, Maltseva OV, Forney LJ (1996) Capture of a catabolic plasmid that encodes only 2,4-dichlorophenoxyacetic acid:alpha-ketoglutaric acid dioxygenase (TfdA) by genetic complementation. Appl. Environ. Microbiol. 62: 2470–2476 [PubMed] [Google Scholar]
- Top EM, Springael D, Boon N (2002) Mobile genetic elements as tools in bioremediation of polluted soils and waters. FEMS Microbiol. Ecol. 42: 199–208 [CrossRef] [PubMed] [Google Scholar]
- Tschäpe H (1994) The spread of plasmids as a function of bacterial adaptability. FEMS Microbiol. Ecol. 15: 23–32 [Google Scholar]
- Van Elsas JD, McSpadden Gardener BB, Wolters AC, Smit E (1998) Isolation, characterization, and transfer of cryptic gene-mobilizing plasmids in the wheat rhizosphere. Appl. Environ. Microbiol. 64: 880–889 [PubMed] [Google Scholar]
- Van Elsas JD, Fry JC, Hirsch P, Molin S (2000) Ecology of plasmid transfer and spread. In Thomas CM, ed, The horizontal gene pool: bacterial plasmids and gene spread, Harwood Scientif. Publ., UK, pp 175–206 [Google Scholar]
- Van Elsas JD, Turner S, Bailey MJ (2003) Horizontal gene transfer in the phytosphere. New Phytol. 157: 525–537 [CrossRef] [Google Scholar]
- Van Elsas JD, Turner S, Trevors JT (2006) Bacterial conjugation in soil. In Nannipieri P, Smalla K, eds, Nucleic Acids and Proteins in Soil, Springer Berlin Heidelberg, pp 331–353 [Google Scholar]
- Vaneechoutte M, Young DM, Ornston LN, de Baere T, Nemec A, Van der Reijden T, Carr E, Tjernberg I, Dijkshoorn L (2006) Naturally transformable Acinetobacter sp. strain ADP1 belongs to the newly described species Acinetobacter baylyi. Appl. Environ. Microbiol. 72: 932–936 [Google Scholar]
- van Overbeek LS, Wellington EMH, Egan S, Smalla K, Heuer H, Collard JM, Guillaume G, Karagouni AD, Nikolakopoulou TL, van Elsas JD (2002) Prevalence of streptomycin resistance genes in bacterial populations in European habitats. FEMS Microbiol. Ecol. 42: 277–288 [PubMed] [Google Scholar]
- Vivian A, Murillo J, Jackson RW (2001) The roles of plasmids in phytopathogenic bacteria: mobile arsenals? Microbiology 147: 763–780 [PubMed] [Google Scholar]
- Watanabe T (1963) Infective heredity of multiple drug resistance in bacteria. Bacteriol. Rev. 27: 87–115 [PubMed] [Google Scholar]
- Widmer F, Seidler RJ, Watrud LS (1996) Sensitive detection of transgenic plant marker gene persistence in soil microcosms. Mol. Ecol. 5: 603–613 [Google Scholar]
- Widmer F, Seidler RJ, Donegan KK, Reed GL (1997) Quantification of transgenic plant marker gene persistence in the field. Mol. Ecol. 6: 1–7 [CrossRef] [Google Scholar]
- Witte W (1998) Medical consequences of antibiotic use in agriculture. Science 279: 996–997 [CrossRef] [PubMed] [Google Scholar]
- Wommack KE, Colwell RR (2000) Virioplankton: viruses in aquatic ecosystems. Microbiol. Mol. Biol. Rev. 64: 69–114 [Google Scholar]
- Zechner EL, de la Cruz F, Eisenbrandt R, Grahn AM, Koraimann G, Lanka E, Muth G, Pansegrau W, Thomas CM, Wilkins BM, Zatyka M (2000) Conjugative-DNA transfer processes. In Thomas CM, ed, The horizontal gene pool: bacterial plasmids and gene spread, Harwood Scientif. Publ., UK, pp 87–174 [Google Scholar]
- Zhang X-X, Lilley AK, Bailey MJ, Rainey PB (2004) The indigenous Pseudomonas plasmid pQBR103 encodes plant-inducible genes, including three putative helicases. FEMS Microbiol. Ecol. 51: 9–17 [CrossRef] [PubMed] [Google Scholar]
- Zhao Y, Ma Z, Sundin GW (2005) Comparative genomic analysis of the pPT23A plasmid family of Pseudomonas syringae. J. Bacteriol. 187: 2113–2126 [CrossRef] [PubMed] [Google Scholar]


